The halogens react with carbon to form commercially important compounds. Few examples of interhalogen compounds are ICl, IBr, BrF, BrCl, ClF. The general molecular formula for interhalogen compounds is AB n, where A is the halogen with higher atomic number and is an odd number. The halogens react with each other to form interhalogen compounds, many of which are binary compounds. The hypochlorous acid produced in this reaction is a strong oxidizing agent. The reaction of chlorine is less vigorous with water. The reaction is highly exothermic and produces oxygen. Hydrofluoric acid is the weakest haloacid. 
The halogens react with hydrogen to form hydrogen halides, which dissolve in water to produce hydrohalic acids. 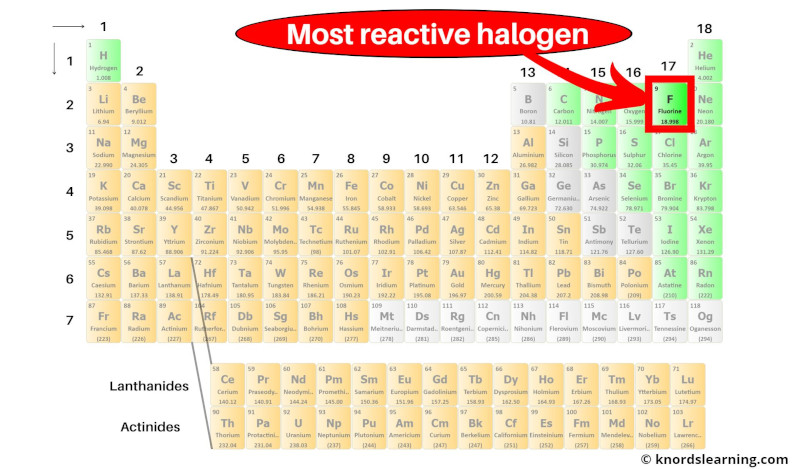
For example, chlorine reacts with iron to give iron chloride and with sodium to give sodium chloride. The halogens react with metals to gain the electron and produce corresponding metal halide. Fluorine is the most powerful oxidizing agent of the group with the most negative electron affinity. The electron affinities of halogens have large negative values thus, the halogens are powerful oxidizing agents. The various properties of halogens are listed in table 1. The valence shell electron configuration of halogens is ns 2 np 5, and they have a tendency to accept an electron to achieve the noble gas configuration. The atomic radius increases from fluorine to iodine. The halogens are not found as single atoms but exist as diatomic molecules. Tennessine is a synthetic element also predicted to be in this group. Astatine is a highly unstable radioactive element, so currently, most of its properties are unknown due to its short half-life. At room temperature, fluorine and chlorine are gases, bromine is a liquid, and iodine a solid. Group 17 elements, known as halogens, are nonmetals. For instance, bromine reacts with fluorine forming bromine trifluoride. Chlorine, however, reacts slower, producing hydrochloric acid and hypochlorous acid - a strong oxidizing agent.Īdditionally, halogens can react with each other, producing binary, covalent compounds called interhalogen compounds. The reaction of fluorine with water is highly exothermic, producing hydrogen fluoride - a strong acid - and oxygen. Halogens react with hydrogen to form polar covalent compounds called hydrogen halides, which often dissolve in water to form hydrohalic acids. For example, chlorine and sodium react to produce sodium chloride, which is an ionic compound. Halogens react with most metals to yield metal halides. It is expressed in kJ/mol, and the negative value indicates the exothermic nature of the reaction. Halogens also exhibit highly negative electron affinities, which is the change in energy when an electron is gained by a neutral atom. Therefore, halogens exhibit high electronegativities with a strong tendency to gain one additional electron to complete their octet, making them highly reactive and powerful oxidizing agents compared to other groups.įluorine is the most potent oxidizing agent amongst halogens and iodine the least. 
In contrast, periodic properties like electronegativity, reactivity, and electron affinity decrease down the group.Įlectron configurations of halogens indicate that they have seven valence electrons and are one electron short of attaining their nearest noble gas configuration. Atomic radii increase with increasing atomic number, and the melting point and boiling point show an upward trend from fluorine to iodine. The physical properties of halogens, such as elemental color, vary moving down the group. Less is known about the properties of the fifth element, astatine, which is a rare and highly radioactive solid. Elemental halogens exist as diatomic molecules.Īt room temperature, fluorine and chlorine are gases, bromine is a liquid, and iodine is a solid. Halogens are a group of five nonmetallic elements – fluorine, chlorine, bromine, iodine, and astatine – belonging to Group 17.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
